GENERAL INFORMATIONName: Helium (he-le-am)
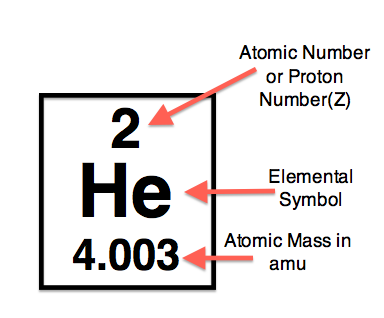
Symbol: He Number on Periodic Table: 2 Atomic Number: 2 Atomic Weight: 4.003Density: 0.1786 g/L Group (Periodic Table): 8 Melting Point: −272.20 °C Boiling Point: −268.928 °C Period (Periodic Table): 1 Description: It is a non metal, colourless, odourless, tasteless, non-toxic, inert and monatomic gas. Discovery: Pierre Janssen, Norman Lockyer 1868 |
ELECTRONS, NEUTRONS, PROTONS
DEFINITION:
Electrons - a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
Neutrons - a subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen.
Protons - a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron.
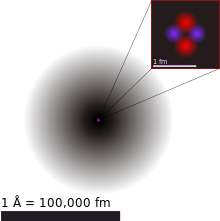
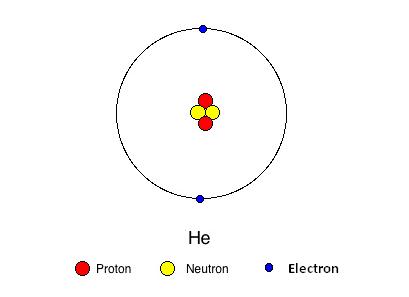
A helium atom is composed of two electrons bound by the electromagnetic force to a nucleus that consists of two neutrons and two protons that are held together by a strong force.
Electrons - a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
Neutrons - a subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen.
Protons - a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron.
A helium atom is composed of two electrons bound by the electromagnetic force to a nucleus that consists of two neutrons and two protons that are held together by a strong force.
HOW WAS IT DISCOVERED?
The first evidence of helium was gathered during a solar eclipse in 1868 by Pierre Janssen who was a French astronomer. He was observing a solar eclipse in India when he noticed the yellow spectral emission lines of the element.
Along with Pierre Janssen an English astronomer, Norman Lockyer observed the same sight. Norman proposed and it was decied to name this element helium after the Greek name for sun (Helios).
Along with Pierre Janssen an English astronomer, Norman Lockyer observed the same sight. Norman proposed and it was decied to name this element helium after the Greek name for sun (Helios).
WHAT IS HELIUM USED FOR?
Helium is used for many things in our everyday life, science, space or medical uses.
- The energy from helium is necessary, for the hydrogen bomb comes from the fusion of hydrogen that transforms into helium.
- A cooling method/medium for nuclear reactors
- An inert gas shied for arc welding
- A gas for supersonic wind tunnels
- A protective gas while growing silicon and germanium crystals or when producing titanium and zirconium.
- Helium is used for MRI and MRE (a blood analysis to determine whether a patient has cancer).
- Pressuring liquid fuel rockets
- Divers and other workers that are in an environment that is under pressure (e.g. submarines, divers) use a mixture of oxygen and helium to create an artificial atmosphere in order to survive.
- Blimps use helium along with filling balloons with it to make they float.
- The energy from helium is necessary, for the hydrogen bomb comes from the fusion of hydrogen that transforms into helium.
- A cooling method/medium for nuclear reactors
- An inert gas shied for arc welding
- A gas for supersonic wind tunnels
- A protective gas while growing silicon and germanium crystals or when producing titanium and zirconium.
- Helium is used for MRI and MRE (a blood analysis to determine whether a patient has cancer).
- Pressuring liquid fuel rockets
- Divers and other workers that are in an environment that is under pressure (e.g. submarines, divers) use a mixture of oxygen and helium to create an artificial atmosphere in order to survive.
- Blimps use helium along with filling balloons with it to make they float.
WHERE AND HOW IS HELIUM OBTAINED?
There are multiple ways to extract and obtain helium: - Helium is present in the gaseous emissions from natural springs
- Present (at 1% level) in natural gas in the USA.
- Helium is obtained from the liquefaction of natural gas.
- Helium is obtained from monazite sands by heating to 1000 degC.
- Present (at 1% level) in natural gas in the USA.
- Helium is obtained from the liquefaction of natural gas.
- Helium is obtained from monazite sands by heating to 1000 degC.
SAFETY HAZARDOUS
Helium is almost harmless and is a gas that is non-toxic but in principle it could asphyxiate through inhalation.